 |
Jasco J-810 Spectropolarimeter coupled to a Peltier and thermal bath
Measures the differential light absorption of asymmetrical molecules in solution.
Applications – Protein secondary structure quantification; provides information of tertiary structure of DNA and proteins; measures protein and DNA stability and charge-transfer transitions. May be used to observe how secondary structures change with environmental conditions or upon interaction with other molecules. Structural, kinetic and thermodynamic information about macromolecules can be derived from circular dichroism spectroscopy.
|
 |
GloMax®-Multi Microplate Multimode Reader
Measures absorbance (UV-Vis), fluorescence and luminescence. It accepts 96-well plates, has dual auto injectors with temperature control and incubation mode.
Applications – Absorbance, static fluorescence and luminescence of DNA, proteins and cell extracts, |
 |
ISS PC-1 fluorimeter coupled to a thermal bath, polarizers, monocromators and filters
Provides information on changes in protein and DNA conformation. It uses a light beam that excites electrons in the sample. Such electrons dissipate this energy emitting light in a higher wavelength.
Applications – Studies on structural behavior of proteins and DNA. It is also used to monitor ligand binding, kinetics and many other interactions. Requires fluorophore that does not interferes with absorption of any component in solution. |
 |
BeckmanCoulter OptimaTM XL-A ultracentrifuge
AUC is one of the most versatile and accurate devices for determining the molecular weight, hydrodynamic and thermodynamic properties of a protein or other macromolecule. It monitors the UV-visible absorbance of the sample while it is under sedimentation in high velocities.
Applications – determine the number of components and species of a sample, the molar mass of each specie and the distribution of sedimentation coefficient |
 |
Dynamic light scattering – cuvette Zetasizer Nano ZS90
DLS or Photon correlation spectroscopy is one of the most popular methods used to determine the size of particles. It is applicable to use in the range from about 0.001 to several microns
Applications – The hydrodynamic radius is one of the main parameters measured by this technique; it gives the size of the molecule in solution and information about oligomeric state. Also, the polydispersity is an important parameter that infers the quality of the sample. This is a good technique to measure stability of samples like proteins and polymers. |
 |
MicroScale Thermophoresis Nanotemper
Microscale Thermophoresis is the directed movement of particles in a microscopic temperature gradient, giving information about any change of the hydration shell of biomolecules due to changes in their structure/conformation results in a relative change of the movement along the temperature gradient and it is used to determine binding affinities.
Applications – Provides interaction analysis of biomolecules. |
|

|
Isothermal Titration Calorimetry – ITC-Vp Microcal
ITC is a thermodynamic technique used for the determination of binding constants, reaction stoichiometry and thermodynamic profile of reactions, which includes enthalpy (ΔH), heat capacity and entropy (ΔS). It is a truly label-free method, no chemical tagging or immobilization of the components is necessary.
Applications – ITC provides a direct measurement of these heats and is particularly well suited for the study of the noncovalent interactions involved in protein – protein and protein – ligand interactions. |
|

|
Differential scanning calorimetry – DSC Microcal
Differential Scanning Calorimetry (DSC) measures the heat changes that occur during controlled increase (or decrease) in temperature.
Applications – DSC can elucidate the factors that contribute to the folding and stability of native biomolecules, including hydrophobic interactions, hydrogen bonding, conformational entropy, and the physical environment. |
|
|
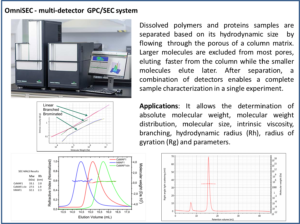
OmniSEC – multidetector GPC/SEC system
Dissolved polymers and proteins samples are separated based on their hydrodynamic size by flowing through the porous of a column matrix. Larger molecules are excluded from the most pores, eluting faster from the column while the smaller molecules elute later. After separation, a combination of light scattering, viscosimeter and refraction index detectors enables a complete sample characterization in a single experiment.
Applications – determination of absolute molecular weight, molecular weight distribution, molecular size, intrinsic viscosity, branching, hydrodynamic radius gyration radius of proteins and polymers. |